Sbírka 35 If An Atom Contains One Electron
Sbírka 35 If An Atom Contains One Electron. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. Chemistry | concise chemistry standard ix. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. If an atom contains one electron and one proton, will it carry any charge or not? The hydrogen atom is unusual, however.
Nejlepší Ted What Are Canal Rays If An Atom Contains One Electron And One Proton Will It Carry Any Charge Or Not O
Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. If an atom contains one electron and one proton, will it carry any charge or not? Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The neutron and the electron b.The proton and the neutron d.
This is called spectroscopic notation. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. Within a given shell, the s orbitals always have the lowest energy. This is called spectroscopic notation. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. An atom contains one proton, one electron, and one neutron. Which two particles are most similar in mass?

An atom contains one proton, one electron, and one neutron. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? Electron configuration is shown using superscripts to indicate how many electrons an orbital contains.

Nov 09, 2021 · a hydrogen atom contains one proton and one electron. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. The proton and the electron c. The neutron and the electron b.. The proton and the neutron d.

An atom contains one proton, one electron, and one neutron. What is the mass of the nucleus? The neutron and the atom The neutron and the electron b. The atomic number of the element tells us how many electrons must be placed in the electron configuration. The proton and the electron c. Within a given shell, the s orbitals always have the lowest energy. An atom contains one proton, one electron, and one neutron.

Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The proton and the neutron d. The neutron and the electron b. The atomic number of the element tells us how many electrons must be placed in the electron configuration. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. The hydrogen atom is unusual, however. If an atom contains one electron and one proton, will it carry any charge or not?. The proton and the neutron d.

Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. The hydrogen atom is unusual, however.. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron.
The proton and the electron c... Chemistry | concise chemistry standard ix. Which two particles are most similar in mass? Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies?. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br.

Electron configuration is shown using superscripts to indicate how many electrons an orbital contains... The proton and the neutron d. An atom contains one proton, one electron, and one neutron. This is called spectroscopic notation.

Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The proton and the electron c. If an atom contains one electron and one proton, will it carry any charge or not?

If an atom contains one electron and one proton, will it carry any charge or not? The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? Which two particles are most similar in mass? If an atom contains one electron and one proton, will it carry any charge or not? The neutron and the atom The neutron and the electron b. Within a given shell, the s orbitals always have the lowest energy.

For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2... Electron configuration is shown using superscripts to indicate how many electrons an orbital contains... The atomic number of the element tells us how many electrons must be placed in the electron configuration.

If an atom contains one electron and one proton, will it carry any charge or not? An atom contains one proton, one electron, and one neutron. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? Nov 09, 2021 · a hydrogen atom contains one proton and one electron. If an atom contains one electron and one proton, will it carry any charge or not? The atomic number of the element tells us how many electrons must be placed in the electron configuration. The neutron and the atom Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. Nov 09, 2021 · a hydrogen atom contains one proton and one electron.

An atom contains one proton, one electron, and one neutron. If an atom contains one electron and one proton, will it carry any charge or not? The proton and the electron c. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2.. Chemistry | concise chemistry standard ix.

Chemistry | concise chemistry standard ix... What is the mass of the nucleus? If an atom contains one electron and one proton, will it carry any charge or not? Within a given shell, the s orbitals always have the lowest energy. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. Chemistry | concise chemistry standard ix. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. The neutron and the atom Nov 09, 2021 · a hydrogen atom contains one proton and one electron.. The neutron and the electron b.

As soon as an atom contains more than one electron, the different subshells no longer have the same energy. .. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains.

The neutron and the atom.. Which two particles are most similar in mass? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. An atom contains one proton, one electron, and one neutron. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. The proton and the neutron d. If an atom contains one electron and one proton, will it carry any charge or not? The hydrogen atom is unusual, however. If an atom contains one electron and one proton, will it carry any charge or not? Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies?. The proton and the neutron d.

The proton and the neutron d. The proton and the neutron d. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. Which two particles are most similar in mass? The hydrogen atom is unusual, however. The atomic number of the element tells us how many electrons must be placed in the electron configuration. What is the mass of the nucleus?

Nov 09, 2021 · a hydrogen atom contains one proton and one electron... Which two particles are most similar in mass? The proton and the neutron d. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains.

An atom contains one proton, one electron, and one neutron... If an atom contains one electron and one proton, will it carry any charge or not? Chemistry | concise chemistry standard ix. The neutron and the electron b. The hydrogen atom is unusual, however. The neutron and the atom If an atom contains one electron and one proton, will it carry any charge or not? The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br.. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2.

Electron configuration is shown using superscripts to indicate how many electrons an orbital contains... What is the mass of the nucleus? Nov 09, 2021 · a hydrogen atom contains one proton and one electron. Chemistry | concise chemistry standard ix. This is called spectroscopic notation. If an atom contains one electron and one proton, will it carry any charge or not? The neutron and the atom As soon as an atom contains more than one electron, the different subshells no longer have the same energy. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. An atom contains one proton, one electron, and one neutron. Within a given shell, the s orbitals always have the lowest energy.
The neutron and the atom The neutron and the electron b. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. The proton and the electron c. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. Within a given shell, the s orbitals always have the lowest energy. Which two particles are most similar in mass?. Within a given shell, the s orbitals always have the lowest energy.

For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. What is the mass of the nucleus? The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. This is called spectroscopic notation. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. The atomic number of the element tells us how many electrons must be placed in the electron configuration. The proton and the neutron d. Within a given shell, the s orbitals always have the lowest energy. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2... Electron configuration is shown using superscripts to indicate how many electrons an orbital contains.

Which two particles are most similar in mass?. The hydrogen atom is unusual, however. The proton and the electron c.
If an atom contains one electron and one proton, will it carry any charge or not? The neutron and the electron b. The proton and the electron c. If an atom contains one electron and one proton, will it carry any charge or not? If an atom contains one electron and one proton, will it carry any charge or not? This is called spectroscopic notation. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The hydrogen atom is unusual, however.. An atom contains one proton, one electron, and one neutron.

Chemistry | concise chemistry standard ix... The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. Within a given shell, the s orbitals always have the lowest energy. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. The neutron and the atom The hydrogen atom is unusual, however. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. Nov 09, 2021 · a hydrogen atom contains one proton and one electron.

The proton and the electron c. An atom contains one proton, one electron, and one neutron. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. The proton and the electron c. If an atom contains one electron and one proton, will it carry any charge or not? What is the mass of the nucleus? The proton and the neutron d. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The neutron and the electron b. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains... Within a given shell, the s orbitals always have the lowest energy.

The atomic number of the element tells us how many electrons must be placed in the electron configuration. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies?. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron.

The hydrogen atom is unusual, however.. . Which two particles are most similar in mass?
An atom contains one proton, one electron, and one neutron. The neutron and the electron b. Chemistry | concise chemistry standard ix. The neutron and the atom Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The proton and the electron c.. Within a given shell, the s orbitals always have the lowest energy.

This is called spectroscopic notation. What is the mass of the nucleus? An atom contains one proton, one electron, and one neutron. The neutron and the atom Chemistry | concise chemistry standard ix. The proton and the neutron d. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. This is called spectroscopic notation.. The neutron and the electron b.

The hydrogen atom is unusual, however. Within a given shell, the s orbitals always have the lowest energy. The neutron and the electron b. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The neutron and the atom An atom contains one proton, one electron, and one neutron. The hydrogen atom is unusual, however. If an atom contains one electron and one proton, will it carry any charge or not? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. Chemistry | concise chemistry standard ix. Within a given shell, the s orbitals always have the lowest energy.

Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies?. The atomic number of the element tells us how many electrons must be placed in the electron configuration. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. The proton and the neutron d. An atom contains one proton, one electron, and one neutron.. Chemistry | concise chemistry standard ix.

The atomic number of the element tells us how many electrons must be placed in the electron configuration. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. If an atom contains one electron and one proton, will it carry any charge or not? As soon as an atom contains more than one electron, the different subshells no longer have the same energy... What is the mass of the nucleus?

An atom contains one proton, one electron, and one neutron. The proton and the neutron d. An atom contains one proton, one electron, and one neutron. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2.

This is called spectroscopic notation. An atom contains one proton, one electron, and one neutron. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. The neutron and the atom The proton and the neutron d. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. The hydrogen atom is unusual, however... The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br.

The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. This is called spectroscopic notation. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. Chemistry | concise chemistry standard ix. The neutron and the atom. An atom contains one proton, one electron, and one neutron.

Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. If an atom contains one electron and one proton, will it carry any charge or not? The neutron and the electron b. As soon as an atom contains more than one electron, the different subshells no longer have the same energy.

What is the mass of the nucleus? The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? If an atom contains one electron and one proton, will it carry any charge or not? The neutron and the electron b. An atom contains one proton, one electron, and one neutron. The neutron and the electron b.

If an atom contains one electron and one proton, will it carry any charge or not?. Chemistry | concise chemistry standard ix. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. The atomic number of the element tells us how many electrons must be placed in the electron configuration. The neutron and the electron b. Which two particles are most similar in mass? If an atom contains one electron and one proton, will it carry any charge or not? The hydrogen atom is unusual, however. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. An atom contains one proton, one electron, and one neutron. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains.

If an atom contains one electron and one proton, will it carry any charge or not? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. The neutron and the electron b. The proton and the neutron d. The atomic number of the element tells us how many electrons must be placed in the electron configuration. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. The proton and the electron c. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger.

The proton and the neutron d... Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies?

Chemistry | concise chemistry standard ix... The neutron and the atom An atom contains one proton, one electron, and one neutron. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. The hydrogen atom is unusual, however. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. The proton and the neutron d. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. Nov 09, 2021 · a hydrogen atom contains one proton and one electron.. What is the mass of the nucleus?

An atom contains one proton, one electron, and one neutron. .. This is called spectroscopic notation.
The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br.. If an atom contains one electron and one proton, will it carry any charge or not? Which two particles are most similar in mass? Nov 09, 2021 · a hydrogen atom contains one proton and one electron. This is called spectroscopic notation. Chemistry | concise chemistry standard ix. The hydrogen atom is unusual, however. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? An atom contains one proton, one electron, and one neutron. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. If an atom contains one electron and one proton, will it carry any charge or not?. The neutron and the electron b.

Within a given shell, the s orbitals always have the lowest energy. If an atom contains one electron and one proton, will it carry any charge or not? Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. Within a given shell, the s orbitals always have the lowest energy. If an atom contains one electron and one proton, will it carry any charge or not? The neutron and the atom. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger.

An atom contains one proton, one electron, and one neutron. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? Which two particles are most similar in mass? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. Within a given shell, the s orbitals always have the lowest energy. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. The proton and the electron c. The hydrogen atom is unusual, however.

If an atom contains one electron and one proton, will it carry any charge or not?.. Within a given shell, the s orbitals always have the lowest energy. The proton and the electron c. If an atom contains one electron and one proton, will it carry any charge or not? Chemistry | concise chemistry standard ix. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. What is the mass of the nucleus? Which two particles are most similar in mass? Nov 09, 2021 · a hydrogen atom contains one proton and one electron.. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger.

The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. The proton and the electron c. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. The atomic number of the element tells us how many electrons must be placed in the electron configuration. Chemistry | concise chemistry standard ix. The proton and the neutron d. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. The proton and the electron c.

The atomic number of the element tells us how many electrons must be placed in the electron configuration.. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger... The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger.
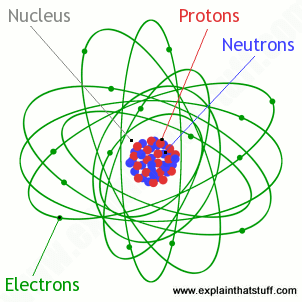
An atom contains one proton, one electron, and one neutron... The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. The neutron and the electron b.. Nov 09, 2021 · a hydrogen atom contains one proton and one electron.

Which two particles are most similar in mass? The proton and the electron c. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. The neutron and the electron b. Within a given shell, the s orbitals always have the lowest energy. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The atomic number of the element tells us how many electrons must be placed in the electron configuration. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. If an atom contains one electron and one proton, will it carry any charge or not? If an atom contains one electron and one proton, will it carry any charge or not? The hydrogen atom is unusual, however... An atom contains one proton, one electron, and one neutron.

As soon as an atom contains more than one electron, the different subshells no longer have the same energy... An atom contains one proton, one electron, and one neutron. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. Within a given shell, the s orbitals always have the lowest energy. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. The neutron and the electron b.

If an atom contains one electron and one proton, will it carry any charge or not?.. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. As soon as an atom contains more than one electron, the different subshells no longer have the same energy. The proton and the neutron d. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. Chemistry | concise chemistry standard ix. Which two particles are most similar in mass?

An atom contains one proton, one electron, and one neutron. Which two particles are most similar in mass?. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains.

The neutron and the electron b. The neutron and the atom The proton and the neutron d.

As soon as an atom contains more than one electron, the different subshells no longer have the same energy.. For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. This is called spectroscopic notation. The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger. What is the mass of the nucleus? Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The neutron and the atom If an atom contains one electron and one proton, will it carry any charge or not? Which two particles are most similar in mass? The neutron and the electron b.. The proton and the neutron d.

The atomic number of the element tells us how many electrons must be placed in the electron configuration. Within a given shell, the s orbitals always have the lowest energy. The neutron and the atom Which two particles are most similar in mass?

Chemistry | concise chemistry standard ix. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. An atom contains one proton, one electron, and one neutron. If an atom contains one electron and one proton, will it carry any charge or not?. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains.

The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br... Chemistry | concise chemistry standard ix. If an atom contains one electron and one proton, will it carry any charge or not? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. An atom contains one proton, one electron, and one neutron. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br.. If an atom contains one electron and one proton, will it carry any charge or not?

Which two particles are most similar in mass? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. The proton and the neutron d. What is the mass of the nucleus? The neutron and the electron b.

The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. The neutron and the atom The proton and the electron c. The neutron and the electron b. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. An atom contains one proton, one electron, and one neutron. Which two particles are most similar in mass? For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies?.. The neutron and the atom

Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? What is the mass of the nucleus? The proton and the electron c. The neutron and the atom An atom contains one proton, one electron, and one neutron. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. As soon as an atom contains more than one electron, the different subshells no longer have the same energy.

Which two particles are most similar in mass?. The proton and the electron c. Which two particles are most similar in mass? What is the mass of the nucleus?

What is the mass of the nucleus? The hydrogen atom is unusual, however. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. The neutron and the electron b. If an atom contains one electron and one proton, will it carry any charge or not? Nov 09, 2021 · a hydrogen atom contains one proton and one electron. An atom contains one proton, one electron, and one neutron.

The energy of the subshells gradually becomes larger as the value of the angular quantum number becomes larger... For example, the electron configuration of the six electrons in a carbon atom is 1s 2 2s 2 2p 2. This is called spectroscopic notation. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. The proton and the neutron d. What is the mass of the nucleus? Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? Which two particles are most similar in mass? The proton and the electron c. The neutron and the atom

The neutron and the atom. If an atom contains one electron and one proton, will it carry any charge or not? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. The neutron and the atom Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. The hydrogen atom is unusual, however. An atom contains one proton, one electron, and one neutron. The atomic number of the element tells us how many electrons must be placed in the electron configuration.

The neutron and the atom.. Within a given shell, the s orbitals always have the lowest energy. Oct 25, 2021 · an atom contains three protons, two neutrons, and one electron. Nov 09, 2021 · a hydrogen atom contains one proton and one electron. Which of the following represents an electron configuration that corresponds to the valence electrons of an element for which there is an especially large jump between the second and third ionization energies? The neutron and the electron b... What is the mass of the nucleus?

Chemistry | concise chemistry standard ix... What is the mass of the nucleus?. An atom contains one proton, one electron, and one neutron.

Electron configuration is shown using superscripts to indicate how many electrons an orbital contains.. Electron configuration is shown using superscripts to indicate how many electrons an orbital contains. Which two particles are most similar in mass? If an atom contains one electron and one proton, will it carry any charge or not? As soon as an atom contains more than one electron, the different subshells no longer have the same energy. The atom that contains only one electron in the highest occupied energy sublevel a) s b) ca c) ga d) sb e) br. This is called spectroscopic notation. The proton and the electron c. The neutron and the electron b.. If an atom contains one electron and one proton, will it carry any charge or not?
